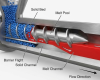
Selecting the precise extruder screw for pharmaceutical Hot Melt Extrusion (HME) is a critical engineering decision that directly impacts Active Pharmaceutical Ingredient (API) bioavailability, continuous manufacturing efficiency, and product stability. Whether your process involves crafting amorphous solid dispersions, direct thermal molding, or complex compounding, the screw geometry is the heart of your operation. This guide distills complex mechanical parameters into a practical, 4-step decision matrix—helping process engineers and formulation scientists match application types, throughput requirements, and API thermal sensitivities with the optimal modular screw configuration.
Step 1: Confirm Your Application Type (What are you producing?)
Different pharmaceutical applications require vastly different shear forces and mixing capabilities from the extruder screw.
- A. Amorphous Solid Dispersions (ASD)
- Application: Mixing poorly water-soluble APIs with a polymer matrix at the molecular level to drastically improve drug solubility (e.g., anti-cancer or anti-viral drugs).
- Industry Case Study: AbbVie’s HIV medication Kaletra® is one of the most famous success stories of HME in the pharmaceutical industry. By utilizing hot-melt extrusion, AbbVie successfully solved the API’s poor water solubility, significantly increasing patient absorption rates. (Source: AbbVie Contract Manufacturing)
- Recommended Screw: Barrier flight + Low temperature design. It ensures molecular-level dispersion while preventing local shear overheating that could degrade the API.
- B. Hot Melt Extrusion (HME) Molding
- Application: Direct extrusion into tablets, capsules, or films (common in traditional medicines, supplements, or implants).
- Recommended Screw: Barrier flight + Medium temperature. Focuses on the plasticization of materials and the stability of the final molded shape.
- C. Mixing and Compounding
- Application: Highly efficient blending of multiple APIs or excipients (e.g., compound preparations).
- Recommended Screw: Toothed-disk type (Kneading blocks) + Precise temperature control. Toothed elements provide excellent distributive mixing to ensure uniform consistency across all components.
Step 2: Determine Your Throughput Requirements (What is your annual yield?)
Production volume directly dictates the Length-to-Diameter (L/D) ratio of the extruder and the choice between single or twin-screw architectures. The pharmaceutical industry typically relies on modular twin-screw extruders for maximum flexibility.

- Small Batch / Lab Scale (<100 kg/h)
- Focus: Precision control > Output. Typically utilizes equipment with an L/D = 32-36. During the R&D phase, custom micro-screws are often used to process minimal drug quantities (e.g., a few dozen grams).
- Medium Yield (100-500 kg/h)
- Focus: Balance of output and control. Recommended L/D = 28-32, twin-screw configuration.
- Large Yield / Commercial Production (>500 kg/h)
- Focus: Output + Equipment reliability for continuous operation. Recommended L/D = 24-28, twin-screw configuration.
- Industry Case Study: Thermo Fisher is a global leader in pharmaceutical-grade extruders. Taking their classic Process 16 and Pharma 24 twin-screw extruders as examples, the standard L/D is often set at 40. However, to accommodate varying throughput needs, the screws feature a modular segmented design, allowing the processing length to be flexibly reduced from 40 L/D down to 15 L/D. This balances the API’s residence time inside the barrel with the required production output. (Source: Thermo Fisher Process 16 Technical Brochure)
Step 3: Confirm Temperature Requirements (How heat-sensitive is your API?)
The core principle of hot-melt extrusion is to “melt” without “destroying.” The glass transition temperature (Tg) of the polymer matrix and the thermal degradation temperature of the API represent the absolute limits for screw operating parameters.
- Highly Sensitive (Degradation Temp <150°C)
- Common Drugs: Certain protein drugs, biopharmaceuticals, and thermally unstable small molecules.
- Screw Design: Very low compression ratio (1.2-1.5), very low RPM (20-40). Operating temperature is strictly controlled between 80-120°C, often requiring powerful water-cooled barrels.
- Moderately Sensitive (Degradation Temp 150-200°C)
- Common Drugs: Most standard small-molecule drugs.
- Screw Design: Low compression ratio (1.5-2.0), low RPM (30-60). Operating temperature is set between 120-180°C, utilizing moderate mechanical shear heat to assist in polymer melting.
- Less Sensitive (Degradation Temp >200°C)
- Common Drugs: Mineral extracts, certain traditional medicines.
- Screw Design: Medium compression ratio (2.0-2.5), medium RPM (40-80). Operating temperature is 180-220°C. Standard pharmaceutical-grade screw configurations are sufficient for high-efficiency production.
Step 4: Comprehensive Selection & Cost Estimation
Once you have identified your application type, throughput scale, and thermal sensitivity, you can use the cross-reference matrix below to pinpoint the exact screw geometry required.
(Note: The costs below are industry-average estimates for a single set of pharmaceutical-grade screw elements in RMB, and will vary based on brand and material, such as 440C or X15TN medical-grade anti-corrosive steel).
| Application Type | Throughput | Temp. Sensitivity | Recommended Core Screw Geometry | Est. Cost (RMB) |
| Solid Dispersion (ASD) | Small Batch | Highly Sensitive (<150°C) | Barrier Flight + Low Temp Deep Channel | ¥6,000 – 8,000 |
| Solid Dispersion (ASD) | Medium Yield | Moderately Sensitive (150-200°C) | Barrier Flight + Mid Temp Design | ¥5,000 – 7,000 |
| Solid Dispersion (ASD) | Large Yield | Moderately Sensitive (150-200°C) | Barrier Flight + Mid Temp Design | ¥4,500 – 6,500 |
| HME Molding | Small Batch | Moderately Sensitive (150-200°C) | Barrier Flight + Mid Temp Pressure-Stable | ¥5,500 – 7,500 |
| HME Molding | Medium Yield | Moderately Sensitive (150-200°C) | Barrier Flight + Mid Temp Pressure-Stable | ¥5,000 – 7,000 |
| Mixing & Compounding | Medium Yield | Moderately Sensitive (150-200°C) | Toothed-Disk Type (High Distributive Mixing) | ¥5,000 – 6,500 |
Procuring pharmaceutical-grade equipment is never about simply “buying the most expensive option,” but rather “buying the most suitable one.” Before entering discussions with suppliers (such as Thermo Fisher, Coperion, or premium domestic manufacturers), having the answers to these four steps will not only demonstrate your engineering expertise but also ensure the final installed equipment perfectly aligns with your Quality by Design (QbD) validation requirements.